“Health care has safety and quality problems because it relies on outmoded systems of work. Poor designs set the workforce up to fail, regardless of how hard they try. If we want safer, higher-quality care, we will need to have redesigned systems of care, including the use of information technology to support clinical and administrative processes”. – Crossing the Quality Chasm: A New Health System for the 21st Century, a consensus report from the Committee on Quality of Health Care in America published by the Institute of Medicine in 2001.
The curious thing to me is how when quality fails, it is often the individual practitioner who takes the fall and pays the price. What Eric Cropp’s story has told me is that the system failed him and failed him many times, but he and he alone owned the error. Eric’s tragic error occurred in 2006 and he was convicted and sent to jail in 2009, 8 years after Crossing the Quality Chasm was published in 2001. Apparently, the health care system has yet to embrace the changes recommended in 2001.
This post is about another potential system failure that could cause harm.
Chronology of Events
November, 2006, The Institute for Safe Medication Practices (ISMP) published a Medication Safety Alert titled “PEN injectors: Technology is not without imPENding risks”. This first warned of impending problems with using this new type of technology.
May, 2008, The Institute for Safe Medication Practices (ISMP) published a Medication Safety Alert titled “Considering Insulin Pens for Routine Hospital Use? Consider this….” This safety alert warned of potential hazards of using these pens, particularly the risk of cross contamination potential if the pens are not properly labeled.
March, 2009, it was reported that 2,114 people may have been put at risk of AIDS and hepatitis by sharing insulin pens and cartridges in two Army hospitals in US.
July, 2010, the FDA published a FDA Consumer Alert titled “Insulin Pens are Not for Sharing”.
May, 2012, The Department of Health and Human Services (CMS) issues a Memorandum to all State Survey Agency Directors titled “Use of Insulin Pens in Health Care Facilities”. Among the discussion recommendations is “Insulin pens must be clearly labeled with patient/resident’s name or other identifiers to verify that the correct pen is used on the correct patient/resident.”
February, 2013, ISMP publishes Medication Safety Alert titled “Hospital Should Reconsider Insulin Pen Use, Says ISMP”.
In July, 2013, The American Society of Health System Pharmacists publishes an article titled “Biological contamination of insulin pens in a hospital setting”. The findings of that article were that of 125 insulin pens tested, 7 tested positive for cells or hemoglobin.
Plenty of evidence that a process change is in order and that a redesigned system of care, including the use of information technology should be developed to support clinical and administrative processes.
Magnitude of the Problem
According to the US Department of Health and Human Services, among hospital acquired infections, blood borne pathogen infections are a significant cause of morbidity and mortality. At any given time, about 1 in 20 inpatients has an infection related to hospital care.
From cdc.gov:
“Applying two different Consumer Price Index (CPI) adjustments to account for the rate of inflation in hospital resource prices, the overall annual direct medical costs of HAI to U.S. hospitals ranges from $28.4 to $33.8 billion (after adjusting to 2007 dollars using the CPI for all urban consumers) and $35.7 billion to $45 billion (after adjusting to 2007 dollars using the CPI for inpatient hospital services). After adjusting for the range of effectiveness of possible infection control interventions, the benefits of prevention range from a low of $5.7 to $6.8 billion (20 percent of infections preventable, CPI for all urban consumers) to a high of $25.0 to $31.5 billion (70 percent of infections preventable, CPI for inpatient hospital services).”
Current System of Care Solutions Fall Short
Floor Stock Dispensing
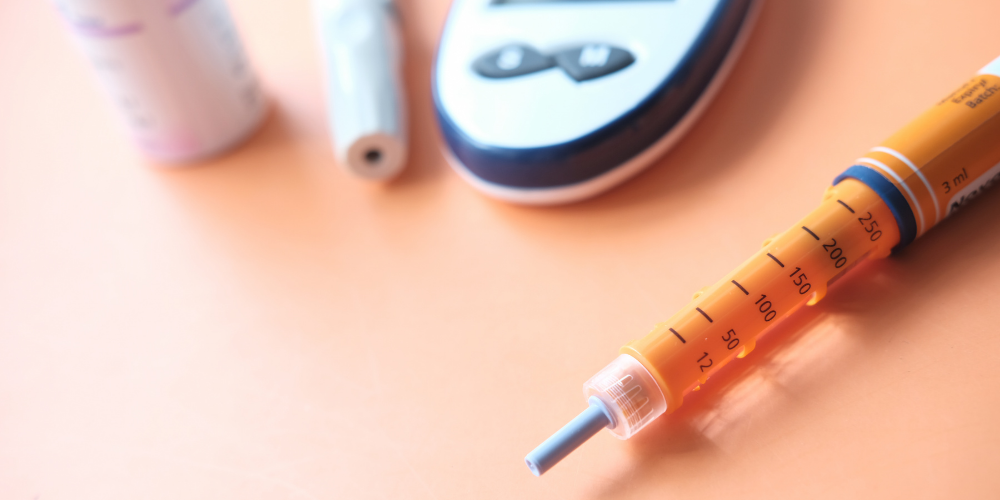
Many institutions will have insulin pens available as a floor stock item in their Automated Dispensing Cabinets (ADC). The problem with this method of drug distribution is having a reliable procedure for the labeling of pens by the nurse at the time of retrieving the pen from the cabinet. I am aware of the label printing capability of the Omnicell system. To utilize this Omnicell capability, it appears that all of the cabinets must be upgraded to the newest Omnicell operating system and that the printing of the expiration date is limited to just one expiration date for all insulin pens.
We understand that the room temperature expiration date is tied to a group of drugs and not to the individual drug. This results in the printing of the shortest expiration date (14 days) for all insulin pens, even an individual pen with a longer expiration date of 28 days must be printed with a 14 day expiration date, resulting in the potential waste of 50% of the drug.
Pharmacy Patient Dispensing
If the insulin pen is dispensed directly from the pharmacy, the pens must be labeled with Patient Name / MRN, room temperature expiration date and the time of dispensing. If this is done by the Pharmacy Operating System, one of the problems is finding space on the Insulin pen to place the label without obstructing important information on the insulin pen label. Placing the label on the removable cap is a patient safety liability in that caps can be interchanged between patients.
This is an important ongoing patient safety issue and we are excited to hear from other health professionals. We invite you to please share your challenges and/or successes with all our visitors. Together we can pool our knowledge, improve the safety of our patients, and protect our health care colleagues.