What is titration and why is it so important for safe medication administration? Chuck DiTrapano, RPh and Maegan Brehm, PharmD explore the significance and objectives of titration schedules, how they impact adverse reactions, and the application of titration to different medication classes.
Dose titration is the administration of a medication over time involving the gradual adjustment of the medication dosage based on response and tolerability. To fully understand titration, we first must explore what it is we are trying to accomplish.
The goal of any therapy is to achieve the optimal therapeutic effect for a specific drug with the minimal number of adverse drug reactions. To achieve that goal, clinicians will often titrate the dose of a drug monitoring therapeutic effect and at the same time the clinical manifestations of side effects.
Every drug will have an optimal dose for a specific patient and at the same time put the patient at risk for any drug related side effect.
What are the ED50 and LD50?
ED50
The ED50 is defined as the effective dose to achieve the desired therapeutic effect in 50% of the population or test subjects. Every drug will have an ED50 that will be personalized based on factors such as patient weight, renal or hepatic function, age, and demographics.
Our goal in providing good pharmaceutical care is to achieve the desired therapeutic effect with the minimal number of side effects.
LD50
The LD50 is defined as the lethal dose, or the amount of drug required to produce toxic levels of the drug in a patient. Every drug also has an LD50 that is personalized based on the same criteria as the ED50.
Our goal in therapy is to achieve ED50 for our patients while staying well below the LD50 to minimize any dose-related adverse drug reactions.
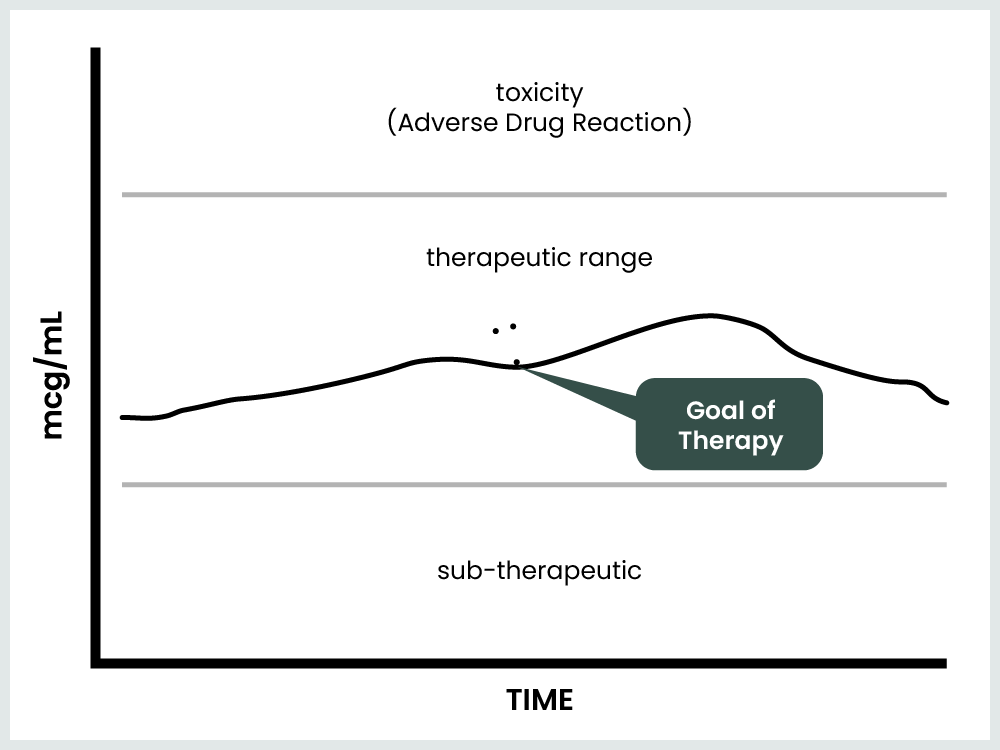
Titrating a dose of a drug can mean an increase or decrease in dose based on where the patient is with regards to effect and toxicity.
Adverse Drug Reactions vs. Adverse Drug Events
Adverse Drug Reactions (ADR) are associated with the pharmacologic aspects of the specific drug, while Adverse Drug Events (ADE) are not specific to the pharmacologic effects of the drug but are more patient-specific, and as a result, less predictable.
Adverse Drug Reactions and Adverse Drug Events are described by The International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use of which the FDA is a member.
Adverse Drug Event is defined as: “Any untoward medical occurrence that may present during treatment with a pharmaceutical product, but which does not necessarily have a causal relationship with this treatment.”
Adverse Drug Reactions are defined as “A response to a drug which is noxious and unintended, and which occurs at doses normally used for prophylaxis, diagnosis, or therapy of disease or the modification of physiologic function.”
Adverse Drug Reactions are further broken down into two primary categories: Type A (intrinsic) and Type B (idiosyncratic).
Type A Adverse Drug Reactions
Type A reactions, (which are most (~85%) adverse reactions) can happen to anyone with a large enough dose. Type A reactions have several specific subcategories.
- Overdose: This occurs when the dose administered is greater than that recommended for your patient.
- Side Effects: Side effects are predictable potential reactions by a patient to a specific drug. An example would be drowsiness caused by diphenhydramine.
- Drug Interactions: When a drug reacts with another drug (prescription or over-the-counter), food, or supplements.
Type B Adverse Drug Reactions
Type B Reactions are not related to the pharmacologic effects of the drug and are thus non-predictable and are more individual to a specific patient. An example of a Type B reaction would be the hypersensitivity reaction associated with drug therapy.
What is the goal of titration?
The goal of titration is to reach the correct patient dose that produces the desired therapeutic effect while minimizing predictable, drug-specific side effects. Since most drugs are dosed by patient weight (mg/kg) or body surface area (mg/m2), we can use standard dosing recommendations to begin therapy.
Titration provides clinicians with the method of achieving the desired therapeutic effect with minimal side effects for a specific patient. Our adjustments may require us to increase the weight-based dose or decrease the weight-based dose.
Titration to Prevent Adverse Drug Reactions.
It is important to provide the overall correct dose for a patient but it is also very important to provide that dose in a way to minimize any adverse drug reactions.
We can use RITUXAN (rituximab) as an example:
Infusion-related reactions are a documented adverse effect of RITUXAN. According to the package insert, these reactions range in clinical severity from urticaria to anaphylactoid reactions. To minimize these risks, patients should be pre-medicated with a corticosteroid and analgesic. Additionally, the infusion should be administered gradually; for a patient’s initial dose, it is recommended to follow a specific titration rate to ensure safety and tolerability.
“Initiate infusion at a rate of 50 mg/hr. In the absence of infusion toxicity, increase infusion rate by 50 mg/hr increments every 30 minutes, to a maximum of 400 mg/hr.”
What is a Fixed Infusion Rate Titration Schedule?
A fixed infusion rate titration schedule is a manufacturer-defined protocol that:
- Starts the infusion at a low initial rate
- Increases the rate at specified time intervals
- Caps the infusion at a maximum allowable rate
- Requires slowing or stopping the infusion if symptoms occur
Unlike ICU titration, these adjustments are not based on physiologic targets such as blood pressure, heart rate, or lab values. Instead, they are based on time and patient tolerance, following a standardized schedule that applies across patients and care settings.
These schedules are most common in biologic therapies and plasma-derived products, especially those associated with infusion-related reactions.
Applying Titration Principles to Specific Infused Medications
Understanding the why behind titration sets the stage for understanding where these principles show up in practice. Many infused therapies, especially biologics and plasma-derived products, use fixed, time-based infusion-rate schedules rather than physiologic titration. These schedules are not arbitrary. They are built into the FDA-approved prescribing information and exist to reduce infusion-related reactions, improve tolerability, and standardize safety across outpatient and home-infusion settings.
Below are the major medication classes where titration schedules are most clinically relevant for infusion teams.
Monoclonal Antibodies (e.g., Rituximab)
Monoclonal antibodies are among the most common medications requiring structured rate escalation. Rituximab is the classic example. Its package insert specifies:
- A low starting infusion rate
- Stepwise increases every 30 minutes
- A maximum allowable rate
- Slowing or stopping the infusion if symptoms occur
These schedules exist because monoclonal antibodies can trigger cytokine release, complement activation, or mast-cell activation, especially during the first infusion. Symptoms such as fever, chills, flushing, dyspnea, or hypotension are more likely when the infusion is started too quickly. By beginning slowly and escalating only if tolerated, clinicians reduce the risk of infusion-related reactions while still delivering the full therapeutic dose.
Subsequent infusions often allow faster titration, but only if the patient tolerated prior doses without significant reactions.
Intravenous Immune Globulin (IVIG)
All IVIG products include explicit infusion-rate tables in their prescribing information. While the exact rates vary by brand, the principles are consistent:
- Start low
- Increase every 15–30 minutes
- Do not exceed the maximum rate
- Reduce the rate if symptoms occur
These limits are essential because IVIG carries risks that are rate-dependent, including headache, renal dysfunction, thrombosis, hemolysis, and aseptic meningitis. Faster infusion rates increase the likelihood of these events.
Fixed titration schedules help ensure predictable, safe administration across infusion centers, ambulatory clinics, and home-infusion programs.
Alpha-1 Proteinase Inhibitors (for Alpha-1 Antitrypsin Deficiency)
Products such as Prolastin-C, Zemaira, Glassia, and Aralast NP are plasma-derived therapies with defined maximum infusion rates. These limits help reduce hypersensitivity reactions, flushing, dyspnea, and headache;effects that become more likely when the infusion is delivered too quickly.
Because these medications are often administered weekly or biweekly in outpatient settings, predictable rate-limited schedules support safe, consistent care without requiring moment-to-moment bedside titration.
Enzyme Replacement Therapies
Many enzyme replacement therapies,particularly those used for lysosomal storage disorders or metabolic enzyme deficiencies,require slow initiation and stepwise escalation. Early infusions carry the highest risk of hypersensitivity reactions, so the first several doses are intentionally slow. As with monoclonal antibodies, subsequent infusions may allow faster rates once tolerance is established.
These therapies are frequently administered in non-acute settings, making standardized titration schedules essential for patient safety and nursing workflow.
Why Titration Schedules Matter
Across all these medication classes, fixed infusion-rate titration schedules are:
- Part of FDA-approved prescribing information
- Considered standard of care
- Essential for minimizing infusion-related reactions
- Critical for safe practice in outpatient and home-infusion environments
Unlike ICU titration (where clinicians adjust rates based on physiologic targets) these medications rely on time-based titration to control exposure, reduce immune activation, and ensure predictable tolerability.
By understanding both the principles of titration and the specific medications that require structured rate escalation, clinicians can deliver safer care, anticipate patient needs, and respond appropriately when infusion reactions occur.
About the Authors:


References:
Iasella CJ, Johnson HJ, Dunn MA. Adverse Drug Reactions: Type A (Intrinsic) or Type B (Idiosyncratic). Clin Liver Dis. 2017 Feb;21(1):73-87. doi: 10.1016/j.cld.2016.08.005. Epub 2016 Oct 13. PMID: 27842776.
Kommu S, Carter C, Whitfield P. Adverse Drug Reactions. [Updated 2024 Jan 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK599521/





